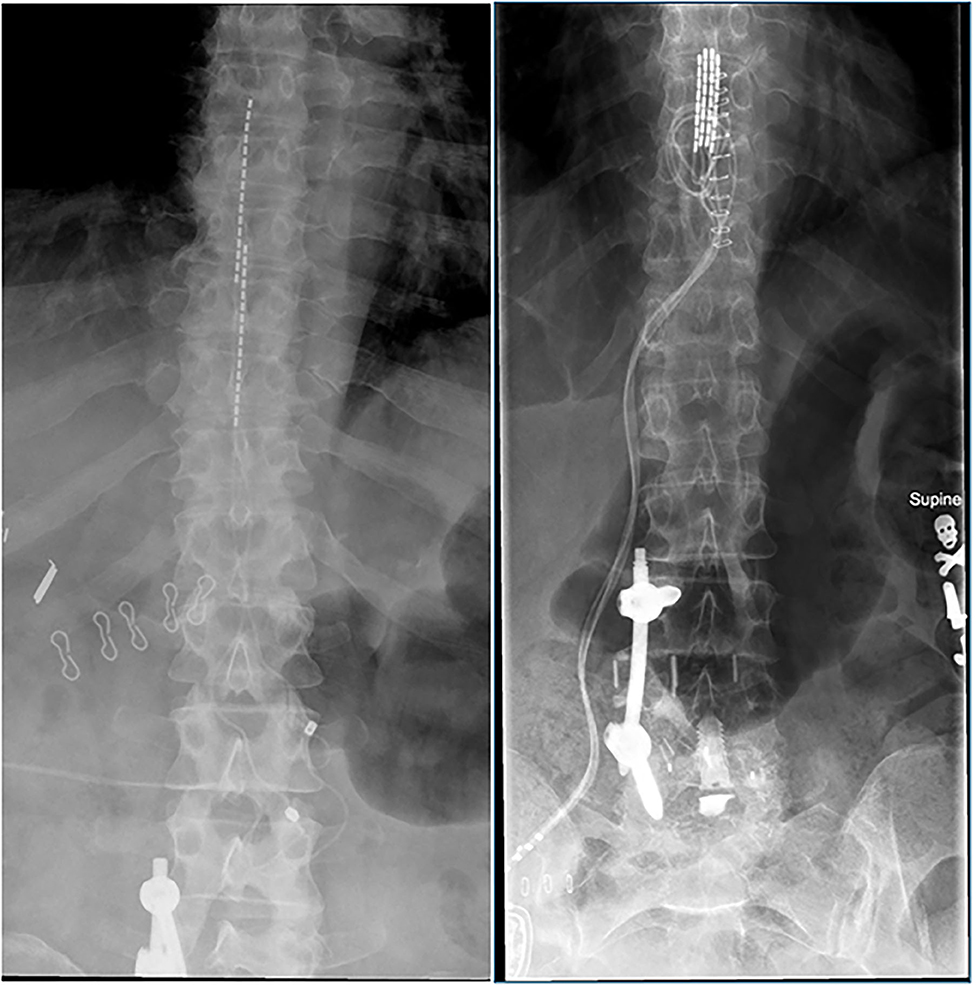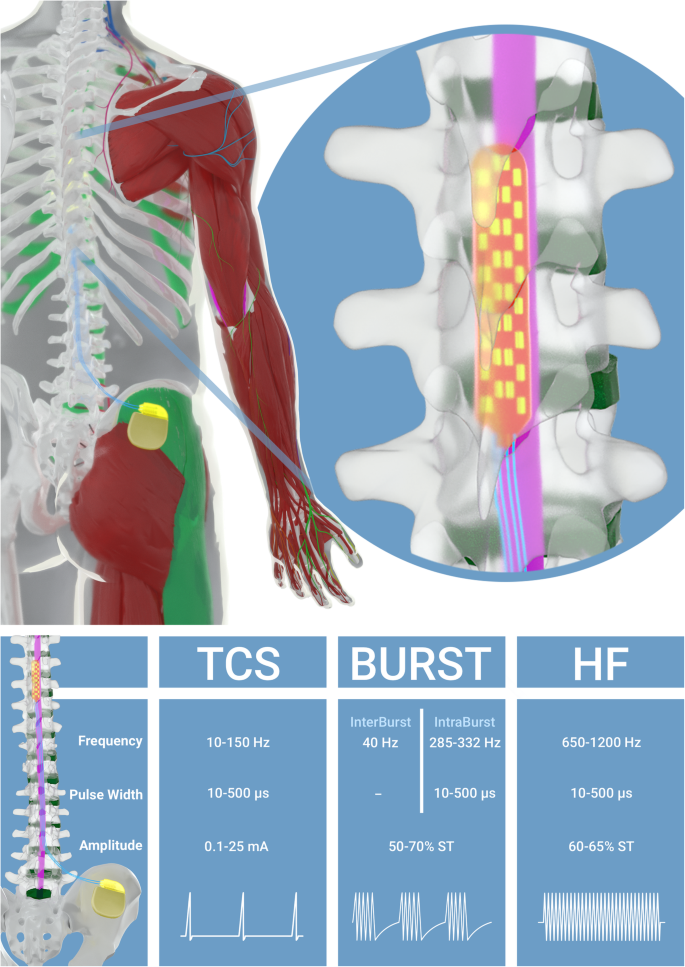
Comparison of conventional, burst and high-frequency spinal cord stimulation on pain relief in refractory failed back surgery syndrome patients: study protocol for a prospective randomized double-blinded cross-over trial (MULTIWAVE study) | Trials

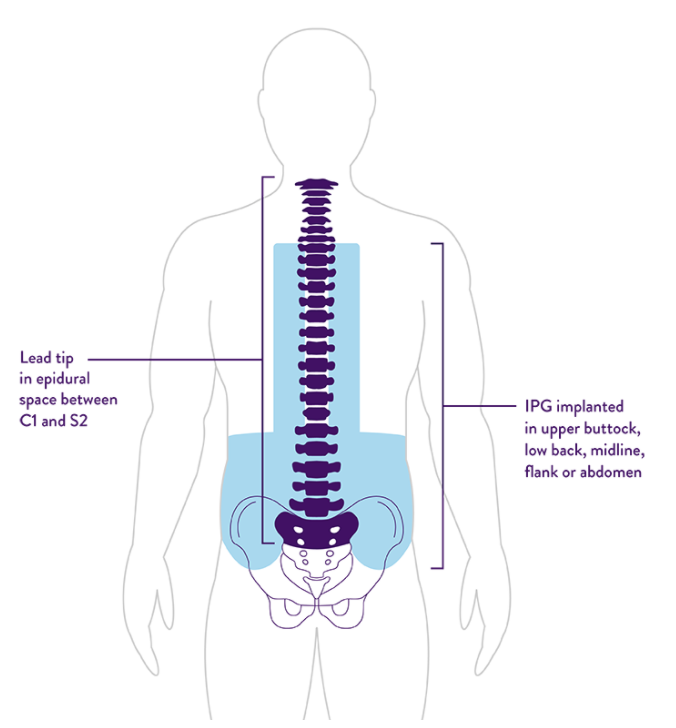
Abbott's Dorsal Root Ganglion Stimulation Outperforms Traditional Spinal Cord Stimulation | Orthopedic Design Technology

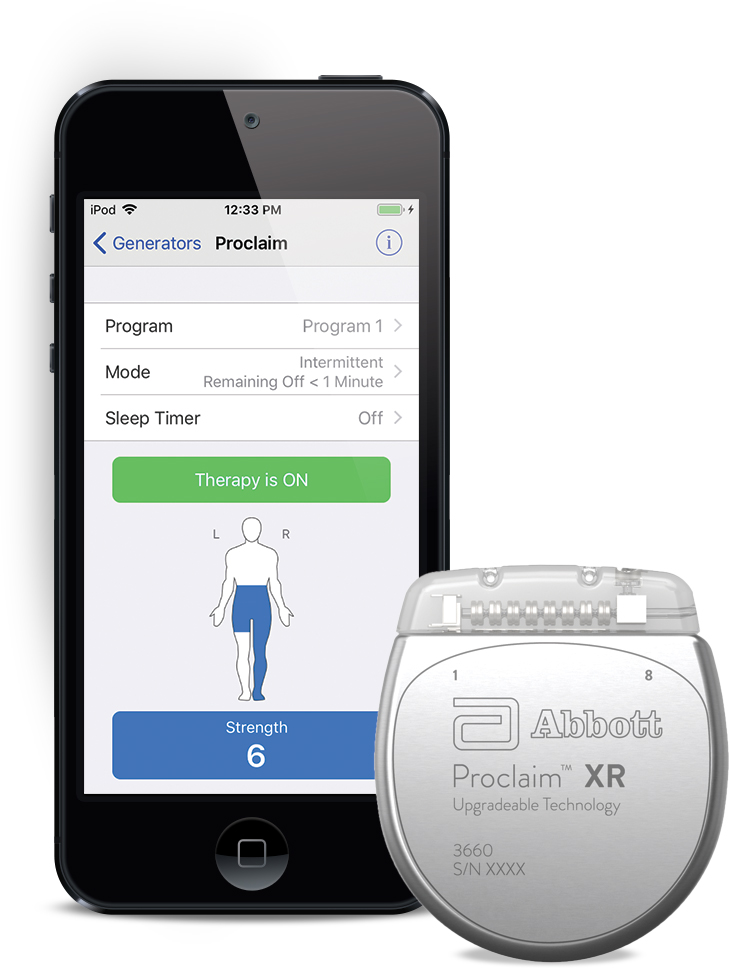
Abbott gets FDA nod for Infinity DBS in Parkinson's disease, unveils positive data on Proclaim XR | BioWorld